|
Graphical Methods for Determining Reaction Order-A Summary Time, Zero-Order Reaction.” The graph shows the plot of concentration versus time for a zero-order reaction. Therefore, if the reaction is zero order, a plot of versus t will produce a straight line with a slope that corresponds to the negative of the product of the rate constant and time, −kt, and a y-intercept that corresponds to the initial concentration, 0 (Figure 17.9 “Concentration vs. Time, Zero-Order Reaction”). The differential rate law for the hypothetical zero-order reaction E → F could be written as: Zero-order reaction rates occur when the rate of reactant disappearance is independent of reactant concentrations. Time, Second-Order Reaction.” The graph shows a plot of versus time for a second-order reaction. Therefore, if the reaction is second order, a plot of versus t will produce a straight line with a slope that corresponds to the rate constant, k, and a y-intercept that corresponds to the inverse of the initial concentration, (Figure 17.8 “ vs. The integrated rate law can be written in the form of a straight line as: The differential rate law can be written: We will examine a reaction that is the latter type: C → D. The rate for second-order reactions depends either on two reactants raised to the first power or a single reactant raised to the second power. We can substitute this data into the integrated rate law of a first-order equation and solve for the concentration after 2.0 years: We are given the rate constant and time and can determine an initial concentration from the number of moles and volume given. If a local factory spills 6,500 moles of this pollutant into a lake with a volume of 2,500 L, what will the concentration of pollutant be after two years, assuming the lake temperature remains constant at 15☌? 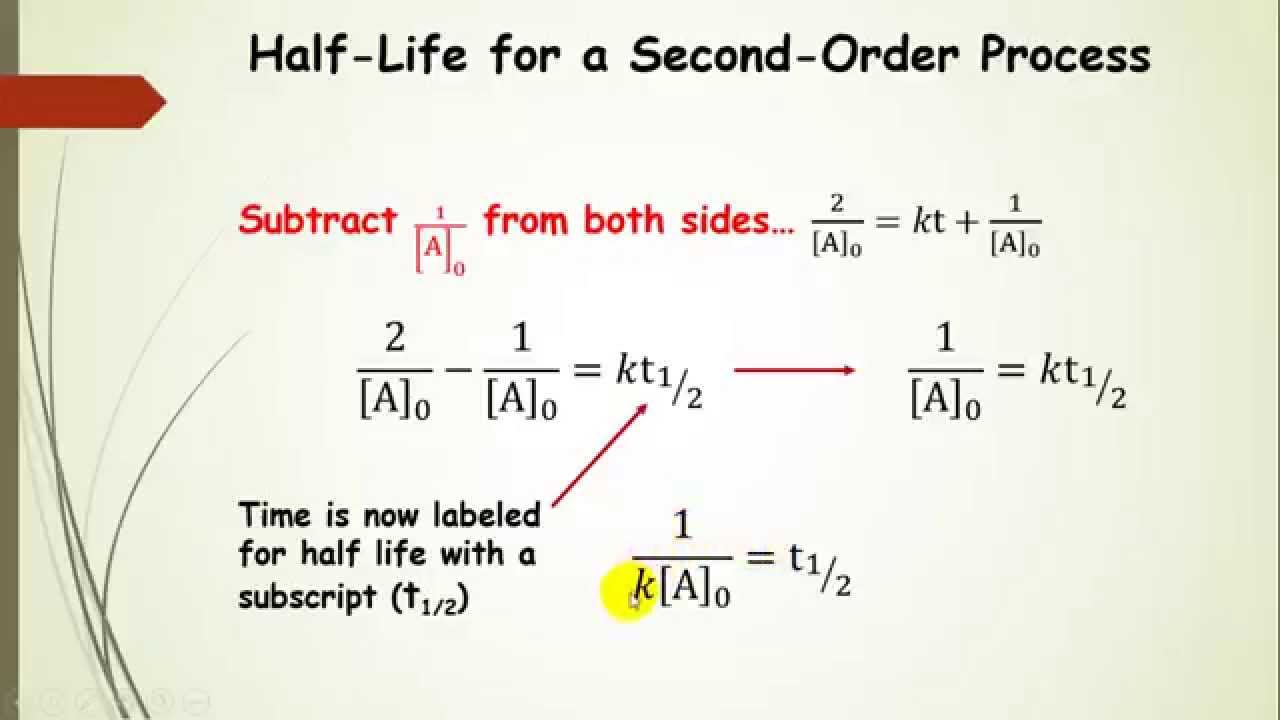
The decomposition of a pollutant in water at 15☌ occurs with a rate constant of 2.39 y −1, following first-order kinetics. “Concentration vs. Time, First-Order Reaction.” This graph shows the plot of the natural logarithm of concentration versus time for a first-order reaction. Therefore, if we were to graph the natural logarithm of the concentration of a reactant (ln) versus time, a reaction that has a first-order rate law will yield a straight line, while a reaction with any other order will not yield a straight line (Figure 17.7 “Concentration vs. Time, First-Order Reaction”). The slope of the straight line corresponds to the negative rate constant, – k, and the y-intercept corresponds to the natural logarithm of the initial concentration. The most useful aspect of the integrated rate law is that it can be rearranged to have the general form of a straight line ( y = mx + b). The process of integration is beyond the scope of this textbook, but is covered in most calculus textbooks and courses. 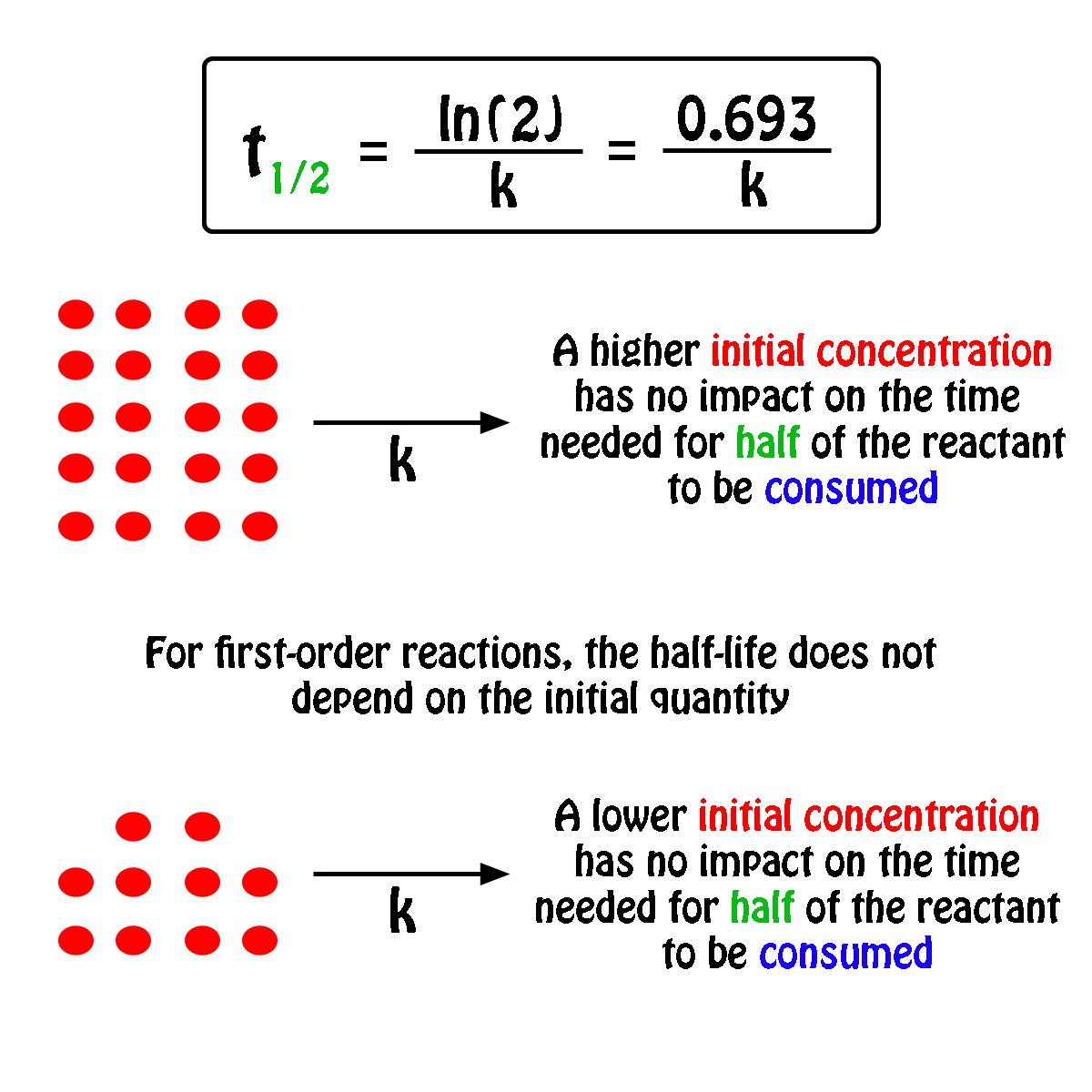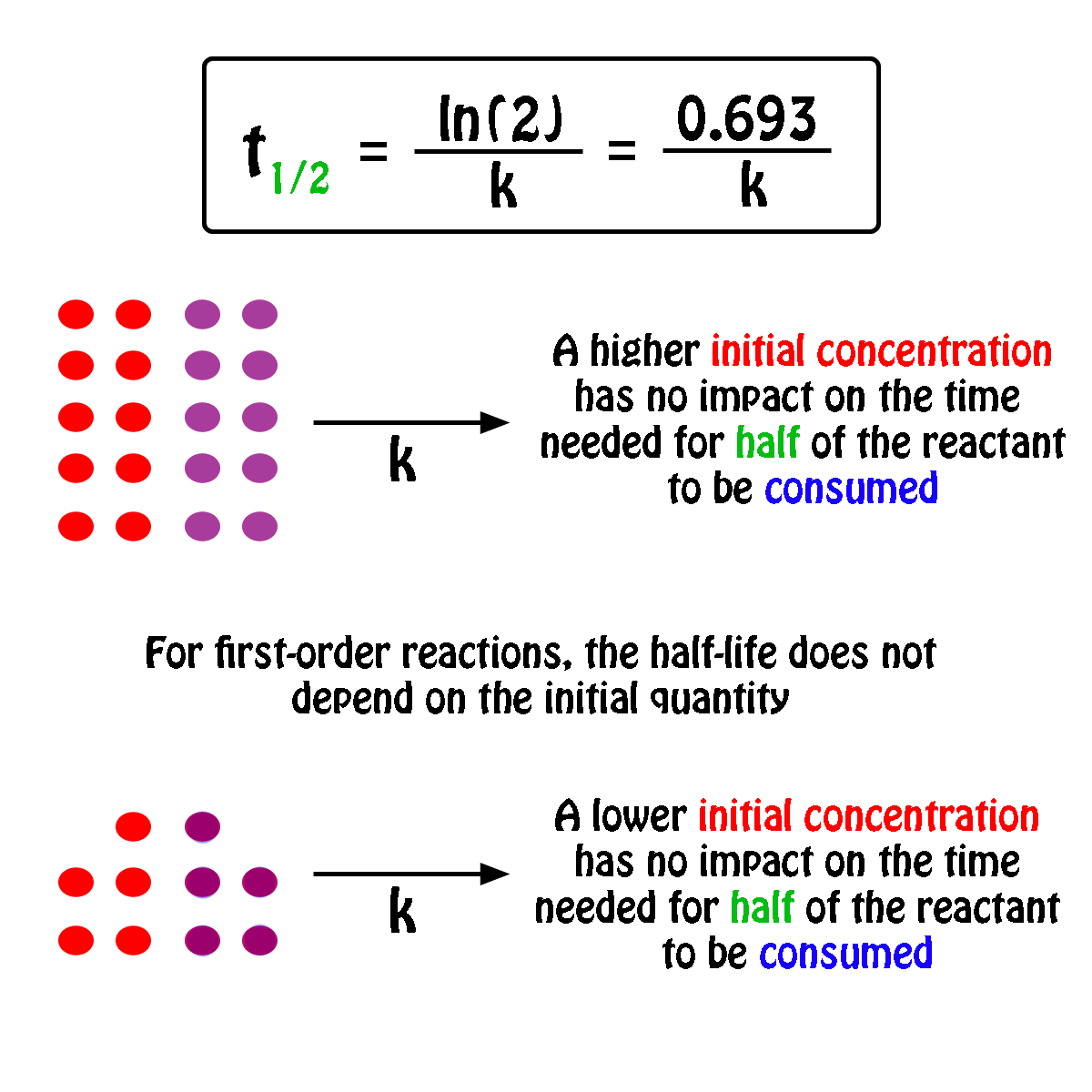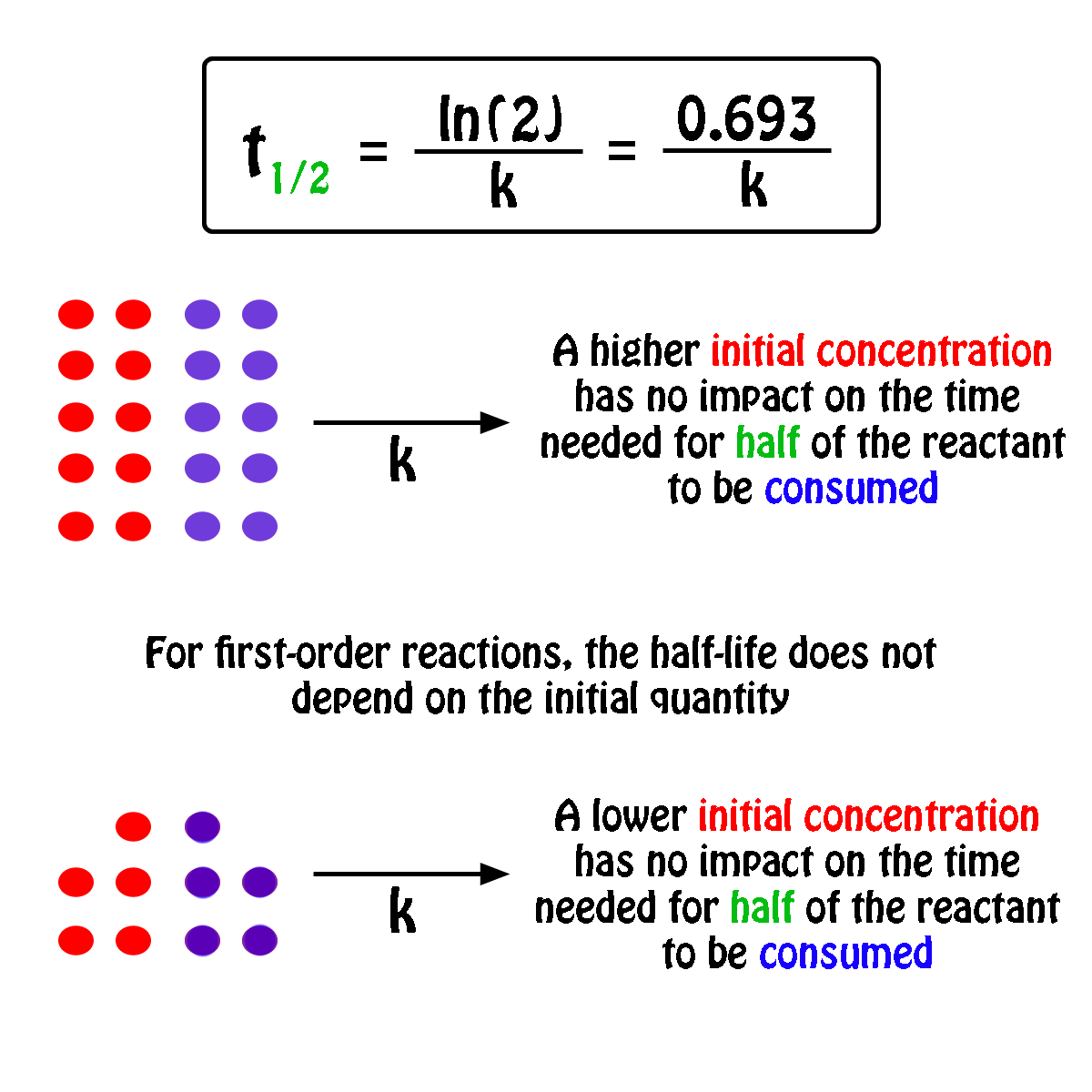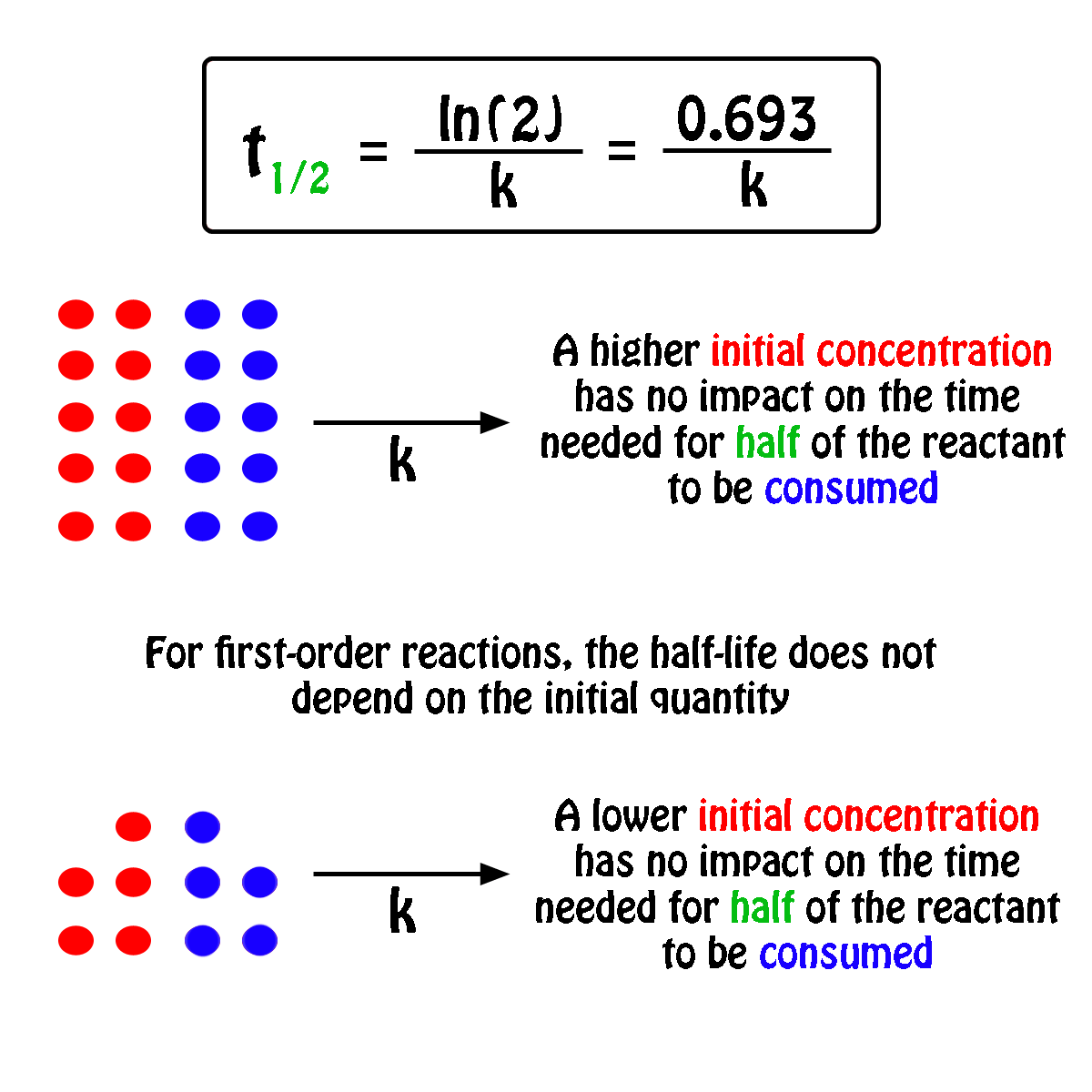
Where “ln” is the natural logarithm, 0 is the initial concentration of A, and t is the concentration of A at another time. We can perform a mathematical procedure known as an integration to transform the rate law to another useful form known as the integrated rate law: This form of the rate law is sometimes referred to as the differential rate law. We have seen earlier that the rate law of a generic first-order reaction where A → B can be expressed in terms of the reactant concentration:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
